
Targeted Gene Delivery
to Treat Cancer
Pioneering cancer therapy to selectively kill tumor cells while sparing healthy tissue
Get in Touch Download One Pager
Pioneering cancer therapy to selectively kill tumor cells while sparing healthy tissue
Get in Touch Download One PagerCytokines and immunotherapies are potent cancer killers. The clinical bottleneck is not efficacy: it is precision delivery to solid tumors.
Current approaches compromise on safety, viral fitness, or tumor penetration. Native cytokines are too toxic systemically. Modified viral coats reduce fitness and scalability. Non-viral delivery lacks amplification and penetration.
Virobix is developing a targeted RNA delivery platform that decouples potency from targeting, enabling safe systemic delivery of therapeutic payloads directly to tumor cells.
De-coupling potency from targeting via a non-modified viral backbone + modular tumor-binding engager
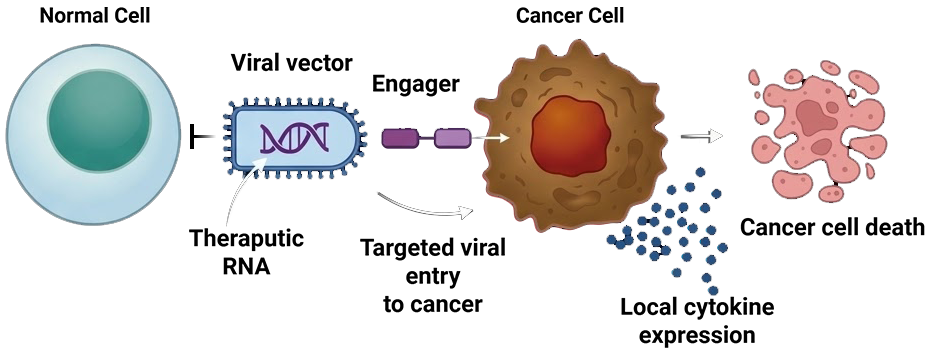
Intravenous administration for full-body reach
No normal tissue sequestration or off-target effects
Coated virus requires receptor-mediated entry
Modular design enables rapid marker switching
From validated proof-of-concept to clinical development
Pancreatic, colorectal, ovarian, and lung cancers. CEACAM6 is a marker for poor prognosis, expressed in 80-90% of late-stage tumors in these indications.
Expansion-ready via modular engager swap. PSMA targeting validated in vitro.
Engager discovery & optimization. In-vitro POC. In-vivo tumor entry. 2 patents filed. External validation.
Virus insert selection & optimization. Engager candidate selection. Biological validation. Early immune activation.
Mechanism & in-vivo PK/PD. Preliminary safety. Organoid efficacy studies. CMC development. Pre-IND planning.
Non-human primate safety. IND filing. First-in-human study (Israel). Phase 1 preparation.
Cytokine and viral experts with proven lab-to-therapeutic execution

PhD Biochemistry, Weizmann Institute. Technology co-inventor. IP commercialization strategist with prior patent licensing success. BD and fundraising lead.





Colab Square startup competition (out of 23 teams)
Selected (170 out of 2,200 applicants worldwide)
Selected for 2026 accelerator program
Technology validated in vitro and in vivo
Interested in learning more about Virobix? We'd love to hear from you.
virobixcompany@gmail.com